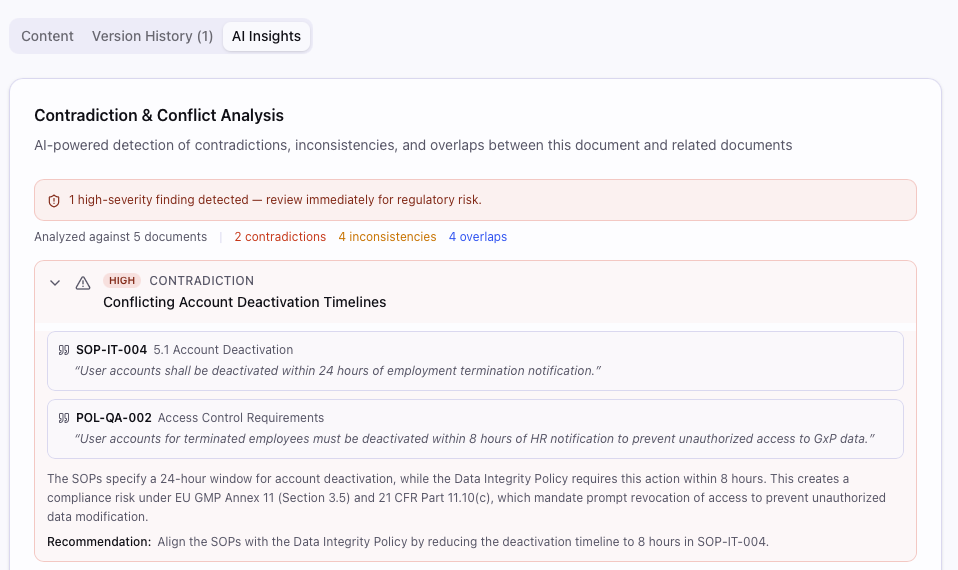
Your budget should fund clinical trials, not systems
QualityMind is the AI-powered eQMS built for European biotech. Full EU GxP compliance — GMP, GCP, GLP. Annex 11 + 21 CFR Part 11 ready. Self-hosted AI — your data never leaves your infrastructure. From €5K/year.